If you have heard that Insight-live does not do glaze chemistry you have heard wrong. It is an ideal tool for exactly this.
Chemistry Calculations Overview
Calculation Type
When a new recipe is first added the chemistry is not shown until you turn it on.
To tell Insight-live to turn on chemistry-display for a recipe: Tap one of the calculation types shown below the recipe (e.g. Formula, Analysis, Mole%).
Recipes remember their calculation-type.

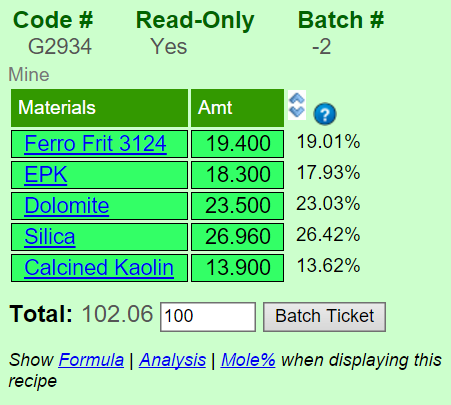
Insight defaults to displaying Auto Unity for the formula. The oxide types are color-coded so that you can quickly cross reference them or direct someone else's attention to a specific one. The formula is always shown, other calculation types are shown in another column to its right (e.g. the percentage analysis is shown here). To redisplay the calculation type options, click the Hide Chemistry link below the chemistry chart.
Unity
Each time the title is tapped (or clicked) Insight-live cycles between the calculation types it can do. They are as follows:
•"Auto Unity" is the first setting when you turn calculation on. If the sum of the RO oxides in the raw formula is higher than half the R2O3 oxides, it is re-totalled so they sum to 1, otherwise the latter (for glazes this normally would unify RO, for clays R2O3).
•"RO Unity" retotals so the fluxing oxides sum to 1. You can configure which oxides participate in unity in Preferences.
•"R2O3 Unity" retotals so the R2O3 oxides sum to 1.
•"Non Unity" does not re-total the formula to unity. The formula numbers thus increase as the recipe total increases. Insight normalizes the formula to create a target formula that is more practical for formula-to-batch calculations (otherwise it becomes a nuisance to supply materials in very fractional amounts to match the target formula).
The unity setting will be remembered for each recipe. However when recipes preview unity is always determined automatically.
Other Calculated Items
Insight-live also calculates ratios, thermal expansion and LOI.
More About Unity
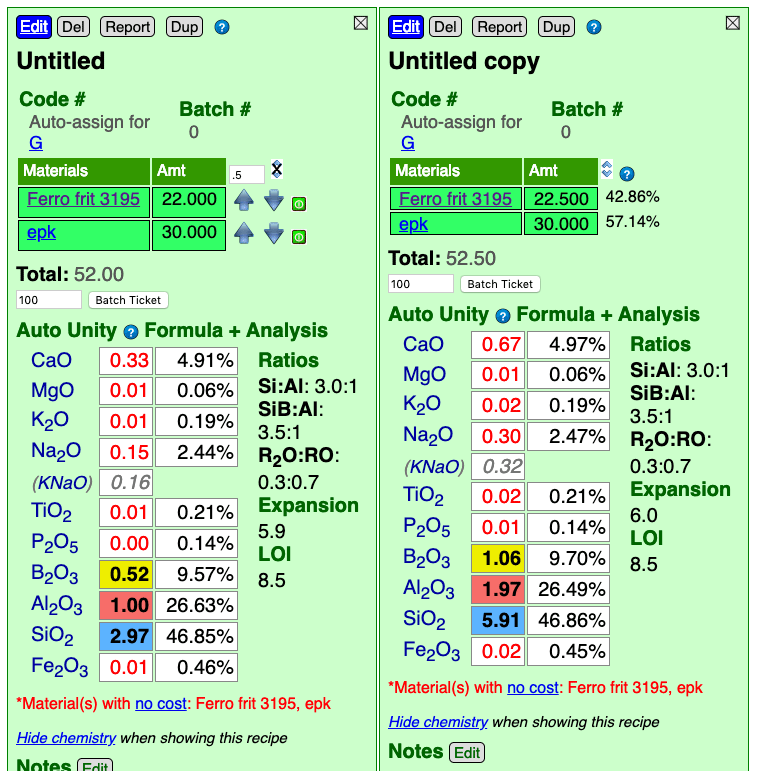
In the side-by-side calculation below note that the second recipe only has 0.5% more Frit, yet the calculated formula is completely different. Why? Because Auto-Unity unifies RO or R2O3 according to which total is greater. On the first one, R2O3 is being unified. On the second, RO. That 0.5% difference was enough to bump it. To fix this, set the second one to RO unity.
More About Calculation Accuracy
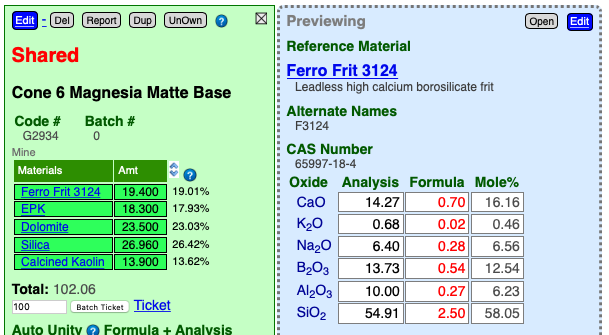
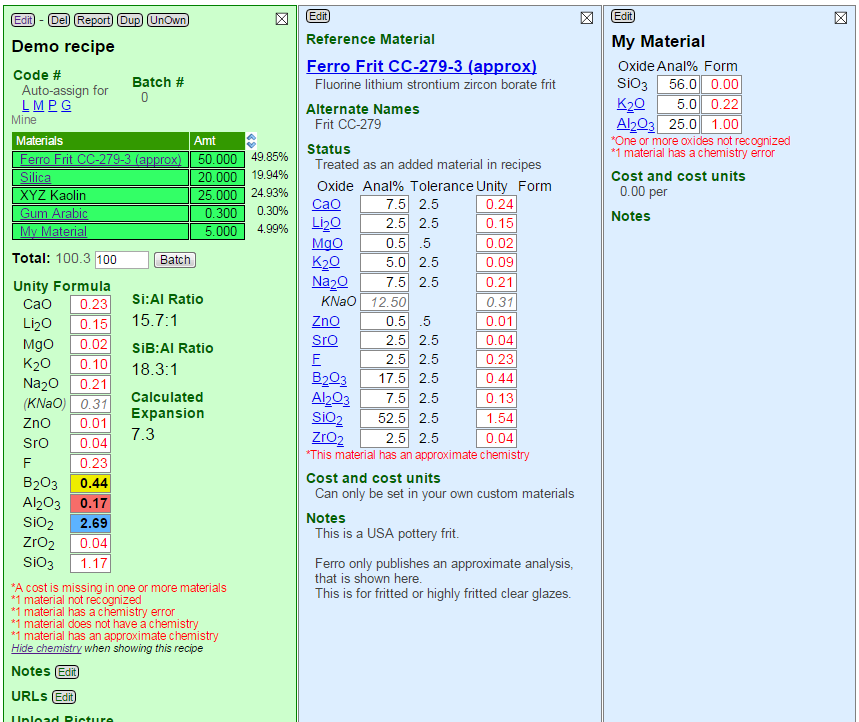
The accuracy of calculated chemistry depends on whether all the materials were found by name among the known ones (reference materials or your own custom ones). You can tell if they were found by noting whether they are hyper-links. Any issues Insight-live spots regarding accuracy will also be noted in red alert notices below the chemistry calculation.
Clicking or tapping a material hyperlink in a recipe opens a blue preview panel showing details about it (see below). This information comes from the Digitalfire Reference Library.
*To see details of how Insight-live does a calculation, add "?showcalc=1" to the URL and press the enter key (if the URL already has a ?variable, remove it and replace it with this).
Entering Recipe Materials Correctly
When searching its reference materials Insight-live recognizes proper names in recipes (e.g. "Potash Feldspar" but not "potspar", "Nepheline Syenite" but no "Neph Sy"). The naming requirements are more strict than they were for desktop Insight to prevent linking to a wrong material. Like desktop Insight, Insight-live allows you to specify a material name that will display in reports and displays of the recipe and a lookup that will be used in the database search.
As Insight-live calculates the chemistry it can encounter many issues that affect accuracy or validity. These issues are foot-noted in red below the chemistry. Each of these will produce a pop-up tip as the cursor passes over it (on a desktop browser) with hints on resolving the problem. Otherwise the ? link brings you to this help topic.
Possible issues with calculation accuracy include:
- One or more materials do not have a cost assigned
Click each to check. Your materials likely reference the built-in materials and you cannot edit those. But you can override parts of their information (like the cost). To do this add a material to your personal or group database (using the Add button in the blue materials manager). Name that material after the reference material, then enter just the cost (it will then override the zero cost in the reference material). - One or more materials in the recipe are not recognized
You can spot these materials because they are unlinked in the recipe. Open the blue materials manager and search the reference materials to get the right names, then edit and update your recipe. - One or more materials has a chemistry error
This applies to materials that you add to your own materials database and reference in recipes. Click each one to preview it on the right and check for invalid oxide names. - One or more materials do not have a chemistry defined
Click each material in the recipe to find which is missing a formula. In some cases this may not be a problem (e.g. for organic materials that burn out and do not affect the chemistry). - One or more materials has an approximate chemistry
Click materials in the recipe to find which one. Often this will be a frit for which the manufacturer only supplies an approximate chemistry. There may be nothing you can do about this except just be aware of it. Or, you may have been given the chemistry of that material in confidence. In that case, create a new material in your private or group materials database and give it the same name (actually you can give it a different unique name also). Then enter that chemistry to override the approximate chemistry in the reference material.
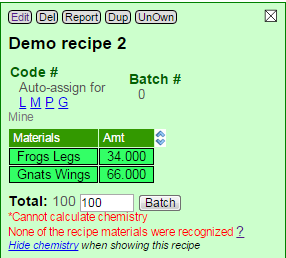
There is a case when Insight-live cannot calculate any chemistry at all. This happens when none of the materials in a recipe are recognized. Fix this problem by researching the proper names in the blue materials manager or by creating your own materials list (to satisfy the material naming in your recipes).
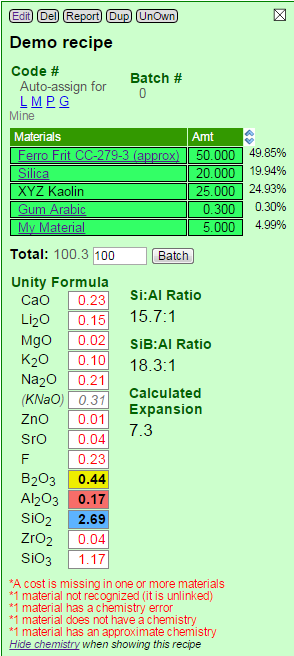
Here is an example of a recipe that has most of the possible errors. Two of the materials are open to demonstrate their specific issues.
Batch to Formula Calculations
More information coming shortly, we are working on this.
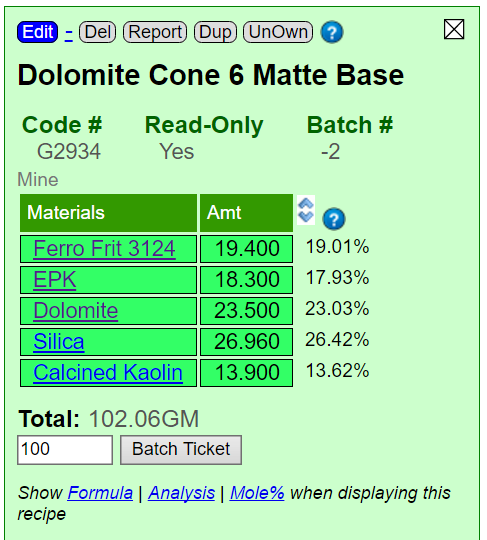
These are automatic in Insight-live: Enter the first material in a glaze batch recipe, turn on calculation for it and from that point on that chemistry will be displayed automatically for that recipe. Forever or until you turn it off.
Correctness of the Calculations
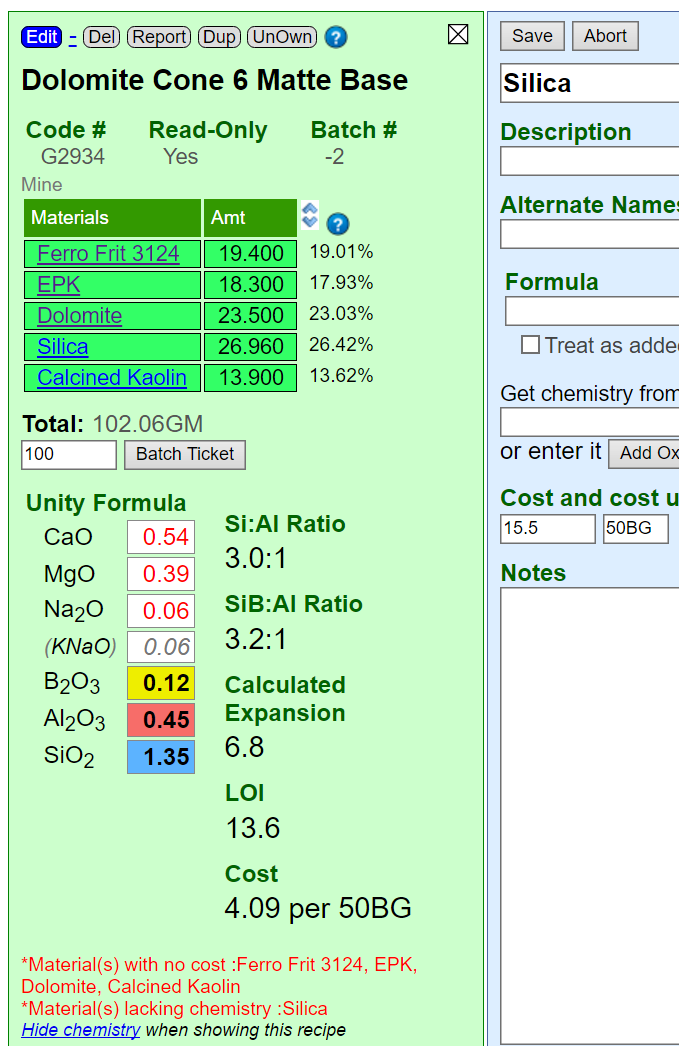
This depends on a variety of factors. Read more about these in the Calculations Overview topic and the Entering Recipes topics.
Here is an example of an incorrect calculation. Although Insight-live is showing the unity formula there are red alerts at the bottom: Silica is lacking chemistry information. That is because we I added our own Silica as a private material (to be able to specify cost). However I did not enter a chemistry for it and neglected to tell it to get the chemistry from the reference material silica (on the "Get Chemistry From" line in the blue panel).
Calculation Mode
Calculation mode automates the nudging of the amounts of individual recipe lines up and down while watching the effect on the chemistry.
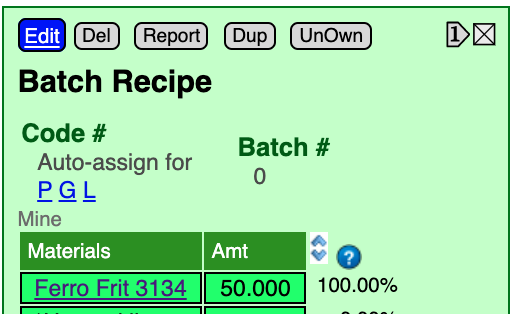
Enter calculation mode by clicking the  )
)
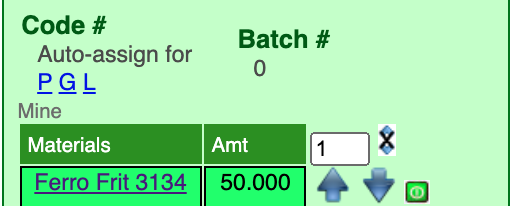
Click the ![]() and
and ![]() arrows to nudge material amounts by the value entered in the blank beside the double-arrows (each time you nudge the page will redisplay and recalculate the chemistry of the recipe).
arrows to nudge material amounts by the value entered in the blank beside the double-arrows (each time you nudge the page will redisplay and recalculate the chemistry of the recipe).
The default nudge amount is "1". Minimize the amount of nudges by changing this as appropriate. An effective strategy is to nudge in one direction until the desired oxide over or undershoots target, then divide the nudge amount in half and proceed in the opposite direction. Repeat until they match.
Terminate calculation mode by clicking  (it is also
is terminated by adding a new material to the recipe (and other actions).
(it is also
is terminated by adding a new material to the recipe (and other actions).
Since multiple recipes can be open side-by-side it is possible to adjust the chemistry of one while comparing it with another. To make it easier to compare specific oxide amounts, Insight-live colors the B2O3 (yellow), Al2O3 (red) and SiO2 (blue).
Comparing the Chemistry of Recipes
Insight-live shows recipes in tall narrow panels. They open side-by-side right-ward. They remember the type of calculation last requested. So just opening multiple recipes automatically enables comparison.
In this video watch how three recipes are opened side-by-side and formula calculation is turned on for each.
You can also watch this at Screencast-o-matic.com
Formula to Batch Calculations
This process is used to derive a recipe of materials that, as a mix, has a desired oxide formula. In Insight-live we do it by setting up two recipes, side-by-side. One is the target-to-be-matched and the other is the batch recipe being built.
Help topic #587 offers a step-by-step lesson to do a formula-to-batch calculation, that may be the most practical way to learn this.
It is important to recognize the limitations of this:
- Theoretically, many different recipes can be calculated to produce the same chemistry, depending on the materials chosen to source each oxide. However in practice, you likely have a limited selection of materials (especially frits). That sometimes means you may have to be satisfied with a less-than-perfect match.
- There may be no combination of materials you want to use that can match the chemistry of your target formula. For example, a formula may demand high enough levels of B2O3, BaO or Li2O that no available frit can supply them without oversupplying other oxides. This can require the need to employ expensive or difficult or toxic raw materials (e.g. Gerstley Borate, lithium carbonate, barium carbonate).
- Theoretically, two different recipes having the same chemistry should fire the same. However in practice, there will be differences in the mineralogy and physics of the materials that also affect melting (e.g. sourcing chemistry from a frit vs. raw materials, from high-melting vs. low-melting raw materials).
- If the accuracy and availability of chemistry information for materials is in question the likelihood of a good match is reduced.
Go to Topic: Formula to Batch Calculations
Converting an Analysis to a Recipe
You can enter the analysis of a material as if it were a recipe. The oxides behave as materials and Insight-live calculates the equivalent recipe.
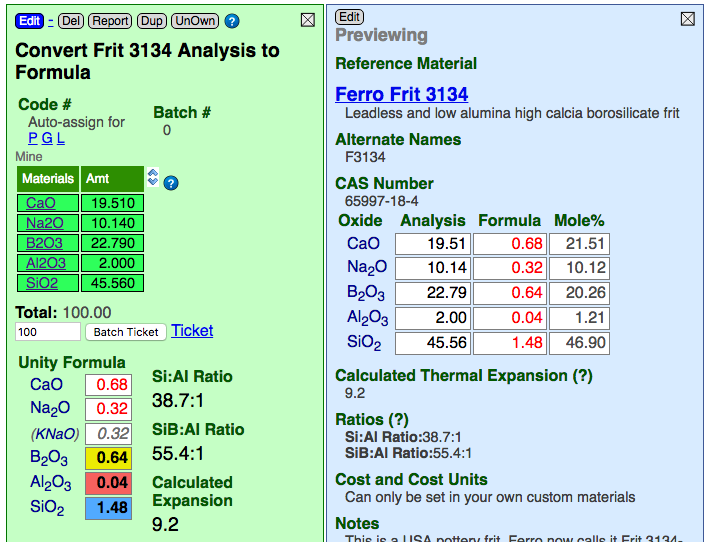
In the example above, Insight-live is calculating the recipe of oxides on the left and the formula of the same material on the right.
How to Enter a Cost for Materials
Obviously, Insight-live cannot put material prices in the master list of materials since these vary with location, country, etc (they have a price, it is set at zero). Insight-live enables you to add your own private materials. When a private material has the name name as a reference material Insight-live uses its cost and chemistry data instead. However private materials can 'refer' to reference ones. That means that Insight will use their names and costs but will get the chemistry from the referred-to material.
Consider the screenshoot below (zoom it to see the detail):
- The Recipe Manager (green title bar and box upper left) with one recipe selected (it is open in the green center panel).
- The Materials Manager (blue title bar on the left) with private (custom) materials added and one selected (it's blue panel is open on the far right).
- The Units Manager (orange title bar lower left) showing the unit abbreviations that have have been defined.
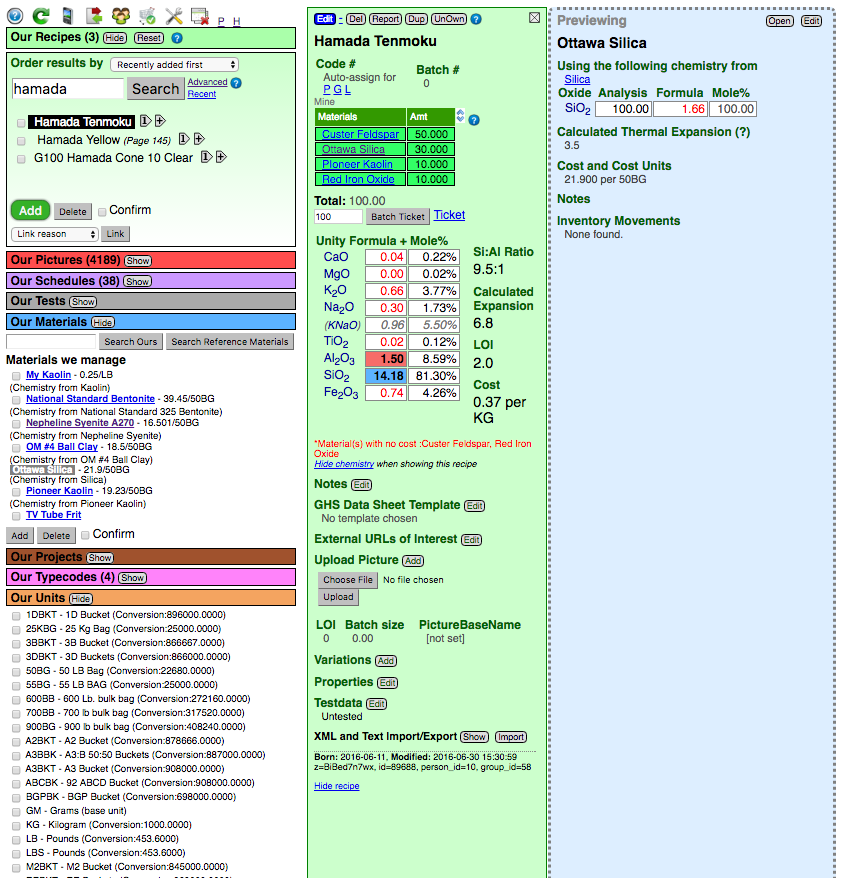
The blue material panel on the right shows "Ottawa Silica". It has been added and is using the chemistry from the reference material "Silica". That means recipes must specify "Ottawa Silica" (if they refer to "Silica" Insight-live will find that in the master list and ignore this cost).

Notice the unit-of-measure has been specified as "50BG" (that is defined in the units manager as 22680 grams, the base unit).

If you do not enter a unit Insight will use the "Cost Units" in your Preferences. If none are specified the default units will be used.
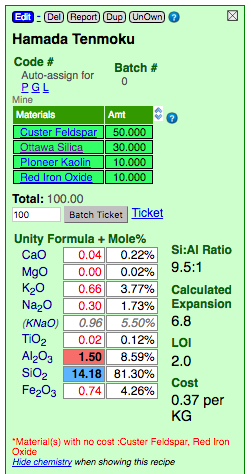
Red footnote warnings appear below the recipe calculations. They warn you about irregularities encountered while doing the math (e.g. materials missing cost information).
Note the warning in this example: "Whiting and Silica have no cost". Why would this happen if these have been added as custom materials and the cost specified for both?
Note that the cost is displayed using the cost units set in Preferences.
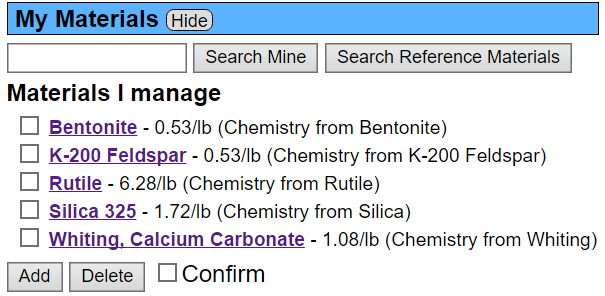
Here is the materials panel. Notice the names: "Silica 325" and "Whiting, Calcium Carbonate". Yet the recipe above refers to them as "Silica" and "Whiting". Silica is being found in the master list because there is a material there precisely matching that name. Whiting, on the other hand, is not in the master list. Insight-live does a second search through the master list when this happens, looking for alternate material name matches (and it successfully finds whiting, but with no cost).
It is not advisable to use the default units, grams, as a cost unit. We recommend kilograms or pounds, check the correct abbreviation in your units manager and specify that as the cost-unit in preferences.
Thermal Expansion, LOI and Ratio Calculations
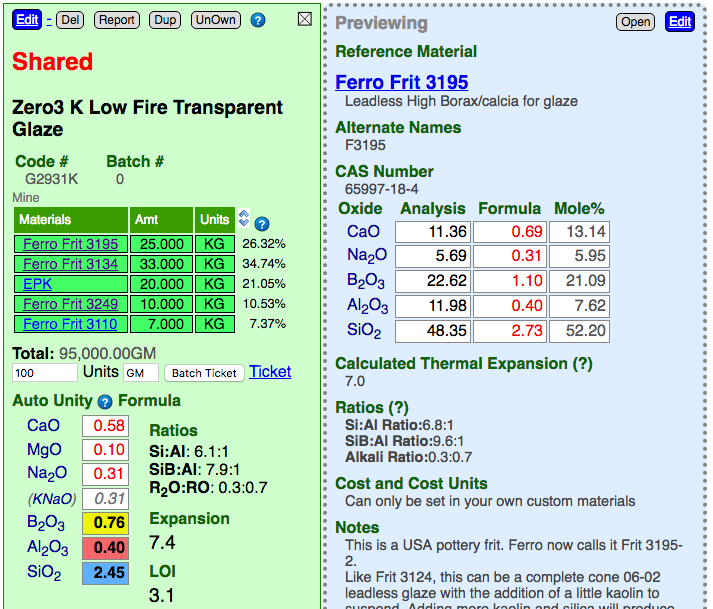
Insight-live calculates the thermal expansion, LOI and various formula ratios for recipes (shown to the right of the chemistry in the green recipe panel) and materials (below the chemistry in the blue material panel). Thermal expansion calculations are based on known expansion values for the oxides (as shown in your Preferences dialog), these are additively accumulated based on the oxide proportions of the currently calculated formula. Unity oxides for the formula are also shown in your Preferences dialog. The LOI and ratio numbers are calculated from the chemistry information in the materials.
A valuable feature of the Insight-live panel system is the ability to click a material in a recipe and see its detail in a panel to the immediate right. In the above example, we can compare the calculated thermal expansion of a material to that of the recipe as a whole to see whether it is lowering or increasing it (likewise for the ratio and LOI). Note that this is not the thermal expansion of the actual material we find in nature, but the calculated expansion of the oxides it contributes to the glaze melt.
To learn more about how and how and why Insight-live calculates these click the titles above the calculated values.
Body thermal expansion: This cannot be calculated. Click the "Expansion" title to learn why.
Recipe Totalling
There are three factors that determine the way Insight-live handles recipe totaling and how percentages are calculated.
Units-of-Measure
If a recipe contains units Insight-live does not recalculate the total with these same units. Units-of-measure are common in factory situations where recipes are large (and use units like 50lb bags, bulk bags, loader buckets). Since these have already been rounded to as even-as-possible units, re-totaling this way is not normally desired. When you print such a mix ticket and specify a total, Insight-live prints a no-units ticket to the total you request (it converts all the units to base units so you can weight the ticket in grams in the lab).
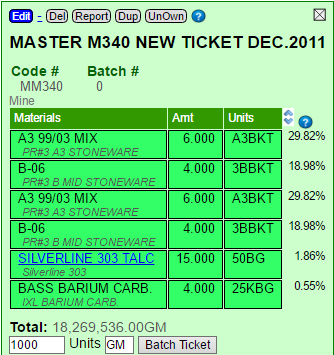 For example, if I print a mix ticket totaling 1000 for this recipe, it will give me 298.2 A3 (the first ingredient).
For example, if I print a mix ticket totaling 1000 for this recipe, it will give me 298.2 A3 (the first ingredient).
Paricipants
When retotaling recipe you can mark specific materials that will not participate in the retotal operation (their amounts will remain unchanged).
Recipes with added materials
Added materials are in the blue section (see below). These usually include colorants, opacifiers, additives. In your Preferences dialog there is a setting for Calculate Percent and Retotal Recipes Based on. You can set this to Full Recipe or Base Recipe. These settings determine how recipes display, print in reports and mix tickets and how they retotal. For the full recipe option it will deem all materials (base and added) as part of a recipe that totals 100. For the latter, the base recipe will total 100 (meaning that percentages shown beside additives are actually of the base-total also). Of course, if you choose the base recipe option and print mix tickets with additives, the total weight of the ticket will be the amount you request plus the additives.
To Retotal a Recipe
- Go into Edit mode by clicking the blue Edit button on the top left of a recipe panel.
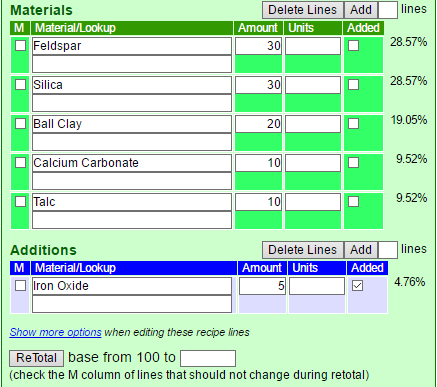
- Check the M column of the lines that should not change during the retotal.
- Enter the new total and click the ReTotal button.
Calculating Firing Temperature of a Glaze
We believe this is not possible. Why? Other software may appear to be able to do this, but they are treating it as a math problem. However it is far more. There are many complex variables that require interpretation skills. Oxides do not just melt as individuals, they interact in complex ways that are well beyond the scope of calculation. The materials that supply those oxides have a wide range of physical and mineralogical properties that affect how they decompose to liberate their oxides to the melt. Another key question is the very definition of the word melt. Some materials melt suddenly, but most soften over a wide range of temperatures. Many glazes are useful across hundreds of degrees, producing a range of visual effects depending on the degree of the melting, melt fluidity, phase sepatations, crystallization and body on which they are applied. Firing methods affect how melting and solidification proceed (e.g. rate of rise, soaking, drop-and-soak, slow cooling). These and other factors make the prediction of absolute values for melting temperature an inexact and highly system-specific science at best.
That being said, frit companies often calculate the softening temperature for their frits. How do they do this? As a relative science. If one frit has a chemistry similar to another then its melting behavior can be predicted in relation to the other if a small change is made. The side-by-side panels make Insight-live a natural for this approach. Technicians know the chemistry needs of the type of glaze they are working with and they know what changes are likely to move the melting temperature in the direction they want without compromising the other needs. The actual fired results, when compared back to the chemistry change(s), build an understanding of the specific 'system' (the temperature, glaze type, the type of chemistry, the materials used, the firing method, etc). Much of our online reference material relates to building skills do this.
Will Insight-live tell me how to change a glaze to make it fire lower? Or higher?
As noted above, work with your glazes and learn to relate their chemistry to the melting behavior (rather than temperature). Develop knowledge within the system of oxides you know and learn the extents to which melting can be controlled and when another oxide system must be partially or fully adopted (new oxides introduced, existing ones reduced or eliminated). For example, fluxes like CaO, MgO that work well at cone 10, are not nearly as effective at middle temperatures. So, it is necessary to add lower temperature fluxing oxides like boron, lithia or zinc. In many cases, a completely different approach is needed. Consider again the task of reducing the firing temperature of a cone 10 glaze to cone 6. Rather than attempting to move its melting range downward it may be better to identify its visual effect mechanisms (opacifiers, variegators, colorants, firing methods, melting fluidity) and transplant that into a base transparent glaze (of the same melt fluidity) that you already have at the desired temperature.
Unexpected Expansion Results
I have glazes with expansions of 6.8 and 7.9 and they both fit the same body. Why?
Expansion calculations are not absolute, they are relative within a system (material or oxide). For example, if you have a dolomite, whiting, feldspar, kaolin, silica glaze and you try a bunch of minor variations, the calculated expansions will give you a fairly accurate indication of which variations have higher and lower expansions and to what degree. But if you make a major change to the oxide balance (perhaps moving toward a high MgO from a high KNaO content) while still using the same materials, the calculated thermal expansion difference between the two will often not accurately reflect the actual measured differences. Even more significantly, if lithium carbonate, for example, is introduced (or a boron frit, or zinc, for example) it is now a different system and different factors affecting the proximity of calculated and actual thermal expansion come into play.
Likely you are employing many systems (which most people are) so there is good reason to think in relative terms. If a glaze is crazing, for example, reduce its thermal expansion and test rather than worrying so much about how much its expansion matches an arbitrary absolute target. Often the degree to which a calculated expansion changes when you adjust the recipe is comparable to the degree to which the actual measured numbers would be different (within a system).
There are other factors that affect the accuracy of thermal expansion prediction also:
- Some oxides, like Li2O do not impose their expansions in a linear fashion or in accordance with their proportion.
- The degree to which a glaze is completely melted determines the degree to which its expansion calculation is valid.
- Crystallization: A melt that freezes to a crystalline solid has a very different thermal expansion than when it freezes to a glass.
- Non melting particles: Zircon imposes its expansion as a non-participant in the glass structure, this is very different than if it were to melt and participate in the glass chemistry. Alumina (calcined or hydrate) is a similar story.
- Oxides interact: The expansion contribution of one may be different depending on which other oxides are present.
Calculating Clay Body Thermal Expansion
Do not do this. Thermal expansion calculations assume a glass where all oxides can impose their proportionate expansions on the whole. This does not work for crystalline solids. Clay bodies do not melt like glazes, the oxides do not form a homogeneous glass, they undergo complex solidification into a mix of amorphous and crystalline solids while cooling in the kiln. A glass and crystal of the same chemistry usually have wildly different physical properties. Variations in particle size distribution, particle mineralogy and shape, firing speed, atmosphere and duration of firing all affect the progressive stages of decomposition and play out of interactions that break and build molecular bonds; these variations are evident in the fired product and all beyond the scope of the chemistry. Consider SiO2 oxide content: It may be equal in two bodies, but one may have most of the SiO2 in quartz grains and the other might have it as a molecular component of feldspar and kaolin. These will, of course, have vastly different thermal expansions.